Islet Cells and Type 1 Diabetes
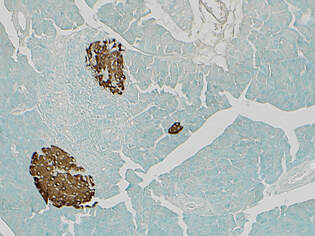
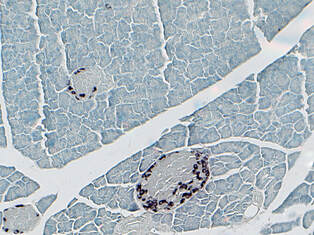
Islet cells are found in the pancreas, and produce hormones responsible for balancing blood sugar levels in the body. There are five unique types of islet cells, two of them being alpha and beta cells. Alpha cells produce glucagon and beta cells produce insulin, both of which play a crucial role in regulating the normal levels of sugar in the blood.
A) How does this affect those with Type 1 diabetes?
Type 1 diabetes is an autoimmune disease that causes the destruction of the individual's islet Beta cells resulting in these cells to produce little to no insulin. This directly impacts their ability to control their blood sugar levels, often resulting in high amounts of sugar in the blood. The most common way of managing type 1 diabetes is to have a patient take insulin injections to make up for what their islet beta cells can't produce, however, islet transplants may also be a viable option for patients. Islet transplants involve taking islets from a donor pancreas, and placing them in the liver of a recipient. The goal of this procedure is to help the recipients regulate the level of glucose in the blood.
B) How does the Rayat Laboratory come into play?
With extensive research, the Rayat Lab aims to find a way to protect islets from harmful effects of hypoxia-induced stress and immune-mediated rejection through the use of natural fruit extracts, genetic engineered islets, and biologic agents (i.e., monoclonal antibodies and small inhibitory molecules) to help those with type 1 diabetes. The Rayat Lab proves that islet transplants are an effective method of treating type 1 diabetes, using fruit extracts to preserve the life of islets. They also showed that genetic engineered islets that express molecules that would stop the body's immune response of attacking the islets after transplantation and a short-term administration of biologic agents to protect the islets from immune-mediated rejection are strategies that allow the islet transplants to survive and produce insulin so that normal blood glucose levels in the recipient’s blood are maintained.
Note: The images below feature two unique immunohistochemistry stains. Each slide features pancreas tissue; the left was stained for insulin and the right for glucagon, both enhanced by methyl green.
Islet cells are found in the pancreas, and produce hormones responsible for balancing blood sugar levels in the body. There are five unique types of islet cells, two of them being alpha and beta cells. Alpha cells produce glucagon and beta cells produce insulin, both of which play a crucial role in regulating the normal levels of sugar in the blood.
A) How does this affect those with Type 1 diabetes?
Type 1 diabetes is an autoimmune disease that causes the destruction of the individual's islet Beta cells resulting in these cells to produce little to no insulin. This directly impacts their ability to control their blood sugar levels, often resulting in high amounts of sugar in the blood. The most common way of managing type 1 diabetes is to have a patient take insulin injections to make up for what their islet beta cells can't produce, however, islet transplants may also be a viable option for patients. Islet transplants involve taking islets from a donor pancreas, and placing them in the liver of a recipient. The goal of this procedure is to help the recipients regulate the level of glucose in the blood.
B) How does the Rayat Laboratory come into play?
With extensive research, the Rayat Lab aims to find a way to protect islets from harmful effects of hypoxia-induced stress and immune-mediated rejection through the use of natural fruit extracts, genetic engineered islets, and biologic agents (i.e., monoclonal antibodies and small inhibitory molecules) to help those with type 1 diabetes. The Rayat Lab proves that islet transplants are an effective method of treating type 1 diabetes, using fruit extracts to preserve the life of islets. They also showed that genetic engineered islets that express molecules that would stop the body's immune response of attacking the islets after transplantation and a short-term administration of biologic agents to protect the islets from immune-mediated rejection are strategies that allow the islet transplants to survive and produce insulin so that normal blood glucose levels in the recipient’s blood are maintained.
Note: The images below feature two unique immunohistochemistry stains. Each slide features pancreas tissue; the left was stained for insulin and the right for glucagon, both enhanced by methyl green.
Below, find attached PDF file of one of Dr. Rayat's presentation on islets and type 1 diabetes. The slides further explores islet transplantation and what Dr. Rayat does in her laboratory.
| louis_d_hyndman_lecture_2005.pdf | |
| File Size: | 9921 kb |
| File Type: | |
Gastric Cancer
Gastric cancer- otherwise known as stomach cancer- is a type of cell formation in the lining of the stomach. The symptoms of this cancer include indigestion/upset stomach, mild nausea, loss of appetite, and heartburn. More advanced symptoms include blood in the stool, vomiting, weight loss, jaundice, and ascites (a buildup of fluid in the abdomen, causing pain). Usually, all these symptoms are noticed after the cancer starts to develop. Physical examinations, blood chemistry studies, a complete blood count, and an upper endoscopy are all different methods used to determine if a patient has gastric cancer.
A) What is neoadjuvant chemotherapy? Can it be used to treat locally advanced gastric cancer?
Currently, there are not enough studies to suggest that certain treatments of gastric cancer are more beneficial than others. Neoadjuvant chemotherapy is one of many methods used to treat cancer, however little is known about its effect on gastric cancer. Neoadjuvant chemotherapy is the administration of medication to a patient with cancer before a main treatment. This form of therapy aims to reduce the size/extent of the cancer, so that the following surgical treatment is easier and more effective. If successful, this procedure significantly reduces a patient's likeliness of receiving further and more extensive treatment for their cancer.- In regards to the treatment of gastric cancer, there is little evidence to show that neoadjuvant chemotherapy reaps many benefits for a patient. However, the Rayat Laboratory aims to use biomarkers, and molecular characterization systems to optimize and show that it is a worthwhile treatment for gastric cancer patients.
B) How is the Rayat Laboratory conducting this research?
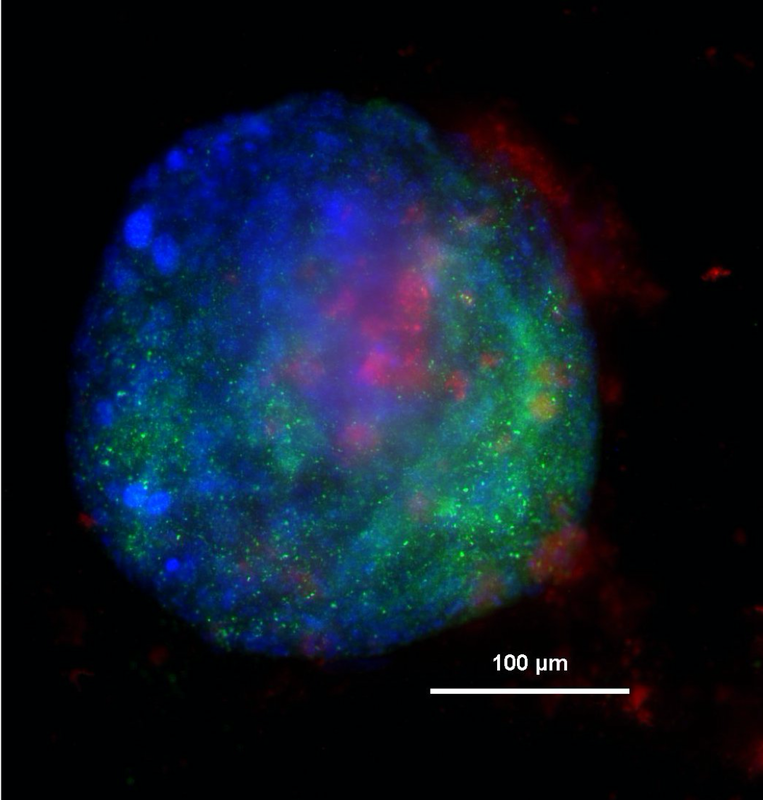
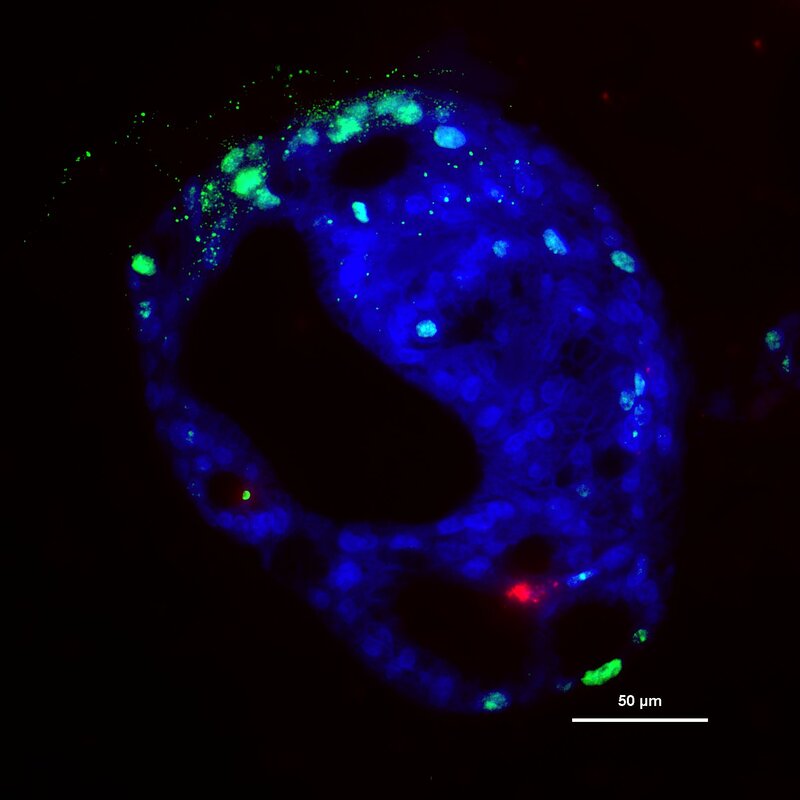
Daniel Skubleny is working with gastric cancer patients who have locally advanced gastric cancer. With their consent, he has been able to utilize cancerous cells from each patient to use in the laboratory setting. Using advanced technology, the Rayat Lab then develops those cells in a three-dimensional space to form miniature organs, or “organoids”, that resemble the patient’s actual cancer. From here, these cells are tested
based on their response to various treatments, one of which being neoadjuvant chemotherapy. With this process, the Rayat Lab in collaboration with Dr. Daniel Schiller hopes to provide evidence to support the efficacy of neoadjuvant chemotherapy in the treatment of gastric cancer.
Note: Seen here is an image of a gastric organoid taken from Daniel Skubleny's lab research.
Mycobacterium tuberculosis:
Tuberculosis (TB), is a contagious bacterial disease caused by Mycobacterium tuberculosis (Mtb) that usually affects the lungs, though it can affect any organ in the body. TB is transmitted from one person to another primarily by aerosol and penetrates the pulmonary alveoli. There it is engulfed by alveolar macrophages and survives within phagosomes. In 90-95% cases, Mtb do not develop the active disease and remain latent in the host for lifetime. Approximately 5-10% of patients with latent infections progress to active disease. Available anti-TB drugs target only a small number of essential functions in the organism and this has proven insufficient to curb the disease due to longer treatment regimen, serious side effects and the emergence of drug resistant strains. In collaboration with Drs. Dennis Kunimoto and Babita Agrawal, the Rayat Lab aims to develop new drug regimens that would complement the existing therapies by providing more effective, less toxic and shorter regimens.
How is the Rayat Laboratory conducting this research?
Drs. Saurabh Garg and Nancy Gupta are working on new molecules with diverse structures different from known drugs. The laboratory aims to evaluate the efficacy, mechanism of action and toxicity of new molecules using cell culture and mouse models of Mtb. The Rayat Lab is also studying resistance development and genome-wide sequencing for the appearance of mutations in Mtb genes in collaboration with Drs. Dennis Kunimoto and
Babita Agrawal.
Credit: Creator: Dr_Microbe | Credit: GettyImages/iStockphoto
Copyright: Dr_Microbe
Tuberculosis (TB), is a contagious bacterial disease caused by Mycobacterium tuberculosis (Mtb) that usually affects the lungs, though it can affect any organ in the body. TB is transmitted from one person to another primarily by aerosol and penetrates the pulmonary alveoli. There it is engulfed by alveolar macrophages and survives within phagosomes. In 90-95% cases, Mtb do not develop the active disease and remain latent in the host for lifetime. Approximately 5-10% of patients with latent infections progress to active disease. Available anti-TB drugs target only a small number of essential functions in the organism and this has proven insufficient to curb the disease due to longer treatment regimen, serious side effects and the emergence of drug resistant strains. In collaboration with Drs. Dennis Kunimoto and Babita Agrawal, the Rayat Lab aims to develop new drug regimens that would complement the existing therapies by providing more effective, less toxic and shorter regimens.
How is the Rayat Laboratory conducting this research?
Drs. Saurabh Garg and Nancy Gupta are working on new molecules with diverse structures different from known drugs. The laboratory aims to evaluate the efficacy, mechanism of action and toxicity of new molecules using cell culture and mouse models of Mtb. The Rayat Lab is also studying resistance development and genome-wide sequencing for the appearance of mutations in Mtb genes in collaboration with Drs. Dennis Kunimoto and
Babita Agrawal.
Credit: Creator: Dr_Microbe | Credit: GettyImages/iStockphoto
Copyright: Dr_Microbe